
Babady NE, Sloan LM, Vetter EA, et al.US Department of Health and Human Services 2022. Tickborne Diseases of the United States: A Reference Manual for Healthcare Providers. Centers for Disease Control and Prevention.Centers for Disease Control and Prevention. Tick-borne Disease, Acute Molecular Panel, Non-Lyme (test code 32338): Includes real-time PCR-based tests for A phagocytophilum DNA (17320) B microti DNA (37314) B miyamotoi DNA (93795) E chaffeensis DNA (11353). 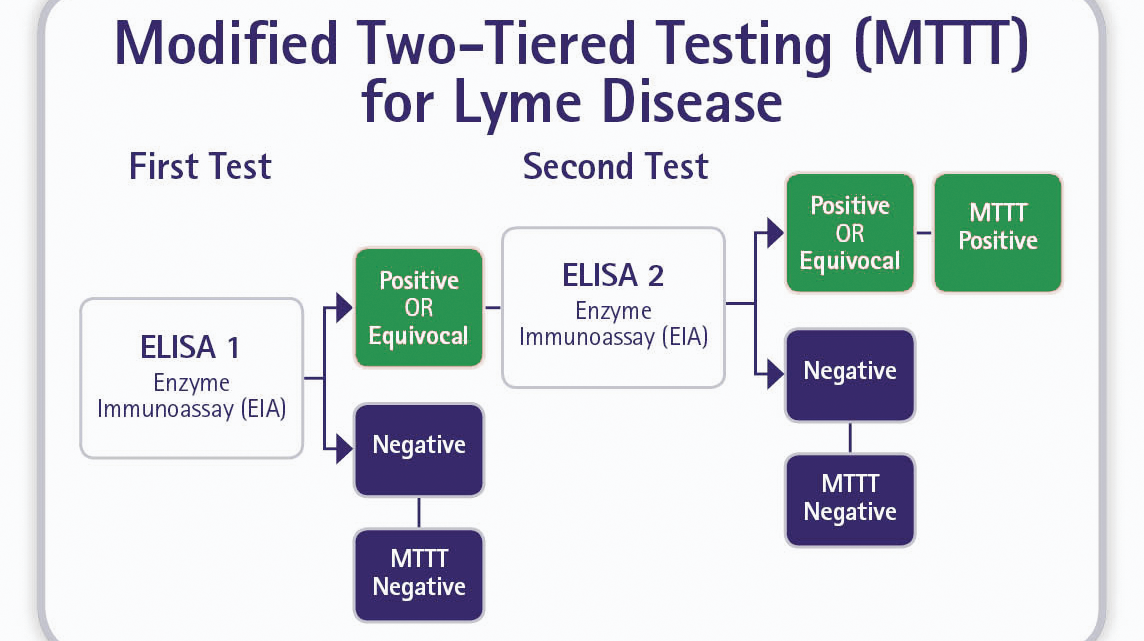
Tick-borne Disease, Acute Molecular Panel (test code 94322): Includes real-time PCR-based tests for A phagocytophilum DNA (17320) B microti DNA (37314) B miyamotoi DNA (93795) E chaffeensis DNA (11353) Lyme Disease ( Borrelia spp) DNA, Blood (15777).Tick-borne Disease, Antibody Panel (test code 36942): Includes A phagocytophilum IgG, IgM (34464) B duncani (WA1) IgG (17231) B microti IgG, IgM (34300) Lyme Disease Ab w/ Reflex to Blot (6646) E chaffeensis IgG, IgM (34271).2,11-13 Quest offers molecular and/or serological tests for B burgdorferi, B miyamotoi, A phagocytophilum, E chaffeensis, and B microti, individually (see Test Directory for individual test codes) and in 3 panels: Molecular tests can be useful in detecting these organisms in early/acute stages of infection, when genetic material from the pathogen can be detected but antibodies remain below the limit of detection (LoD) of serologic assays. 2,11-13 Several studies on co-infected ticks indicate up to 20% of Ixodid ticks can be coinfected with Borrelia burgdorferi and 1 or more other tick-borne human pathogens. Ixodes scapularis (deer) ticks can also carry other human pathogens that have a high degree of symptom overlap, including Borrelia miyamotoi, Anaplasma phagocytophilum, Ehrlichia chaffeensis, and Babesia microti. MTTT: Lyme Disease Antibody with Reflex to Immunoassay (IgG, IgM) (test code 39733)Ĭlick here for a list of additional tests available from Quest for tick-borne infectious diseases.STTT: Lyme Disease Ab with Reflex to Blot (IgG, IgM) (test code 6646).Quest offers test options for both the STTT and the MTTT algorithms: 4 The MTTT algorithm is now considered an acceptable approach for the serologic diagnosis of Lyme disease and may be able to help identify early Lyme disease within the first 30 days of infection. However, on July 29, 2019, the US Food and Drug Administration (FDA) cleared several Lyme disease serologic assays with new indications for use, which allowed an EIA (rather than immunoblot assay) to be used as the confirmatory test in a modified 2-tier testing (MTTT) algorithm. In the standard 2-tier test (STTT) algorithm, a Western blot or immunoblot assay is used for confirmation. 4 When laboratory diagnosis is indicated, current recommendations include using a 2-tier testing approach that begins with a sensitive enzyme immunoassay (EIA), followed by a confirmatory immunoassay for specimens yielding positive or equivocal results. Quest offers testing in accordance with the Centers for Disease Control and Prevention (CDC) guidelines for early/acute Lyme disease testing.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
